“Why Pt Survives but Pd Suffers From SOx Poisoning” Published in JPC Letters
By Giorgina Paiella
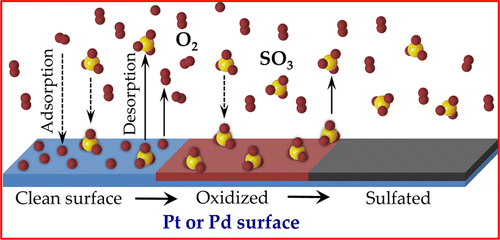
Sulfur in automotive and power plant fuel poisons catalyst surfaces by creating a sulfate layer, thus making the catalysts inactive for emissions mitigation
Graduate student Hom Sharma (Department of Chemical Engineering) and co-authors Dr. Vinit Sharma and Professor Rampi Ramprasad (Department of Materials Science and Engineering), in conjunction with Dr. Ashish B. Mhadeshwar, presently with ExxonMobil Corporation, have recently published their paper, “Why Pt Survives but Pd Suffers From SOx Poisoning,” in the Journal of Physical Chemistry Letters.
It is widely known that the combustion of gasoline in automobiles produces harmful emissions, including carbon monoxide, nitrogen oxides, and unburned hydrocarbons. Environmental regulations require that these harmful emissions be capped at a given limit, which is achieved by converting most of the harmful emissions into harmless gases. Catalysts like platinum (Pt) and palladium (Pd) aid this conversion process. Although palladium is much cheaper than platinum, palladium is poisoned by sulfur oxides (SOx) present in the emissions, while platinum is not. Sulfur poisoning is also referred to as sulfation, a process that produces palladium sulfate (PdSO4) from the reaction between palladium, SOx, and oxygen.
“Why Pt Survives but Pd Suffers From SOx Poisoning” addresses the puzzle of why palladium undergoes sulfation while platinum resists it. The authors use computational quantum mechanics and statistical thermodynamics to explain the intriguing difference between these two catalysts, including the different characteristics that they display under given conditions. The authors also identify key descriptors that correlate to heightened sulfation tendencies, which will prove highly valuable in future research on cheaper, sulfur-resistant catalysts.
Published: April 3, 2015
Categories: faculty, news, publications, research, undergraduate students
Available Archives