“Visualizing Osteogenesis In Vivo Within a Cell–Scaffold Construct for Bone Tissue Engineering Using Two-Photon Microscopy” Contributes Cover Image to Tissue Engineering C
By Giorgina Paiella
 MSE Ph.D. student Max Villa (M.S.), in conjunction with Dr. Liping Wang (M.D.), Dr. Jianping Huang (M.S.), Dr. David W. Rowe (M.D.), and MSE Professor and Associate Dean for Research and Graduate Education Dr. Mei Wei, have jointly published an article in the biomedical journal Tissue Engineering. The multidisciplinary journal features innovative research and applications of tissue growth and regeneration, uniting the principles of engineering and life sciences within the realm of tissue development and regenerative medicine.
MSE Ph.D. student Max Villa (M.S.), in conjunction with Dr. Liping Wang (M.D.), Dr. Jianping Huang (M.S.), Dr. David W. Rowe (M.D.), and MSE Professor and Associate Dean for Research and Graduate Education Dr. Mei Wei, have jointly published an article in the biomedical journal Tissue Engineering. The multidisciplinary journal features innovative research and applications of tissue growth and regeneration, uniting the principles of engineering and life sciences within the realm of tissue development and regenerative medicine.
The article, “Visualizing Osteogenesis In Vivo Within a Cell–Scaffold Construct for Bone Tissue Engineering Using Two-Photon Microscopy” was featured in the Part C: Methods section of the November 2013 section of Tissue Engineering. An optical section of a mouse skull revealing osteoprogenitor cells acquired during a live animal imaging session utilizing two-photon microscopy was featured on the cover.
The research project and paper focus on cell-based bone tissue engineering. While bone can regenerate itself in the case of small fractures, a more severe bone injury requires a bone graft. Presently, the best available graft is a patient’s own bone, but if a patient is missing a significant amount of bone, this method simply does not supply enough to fill the void.
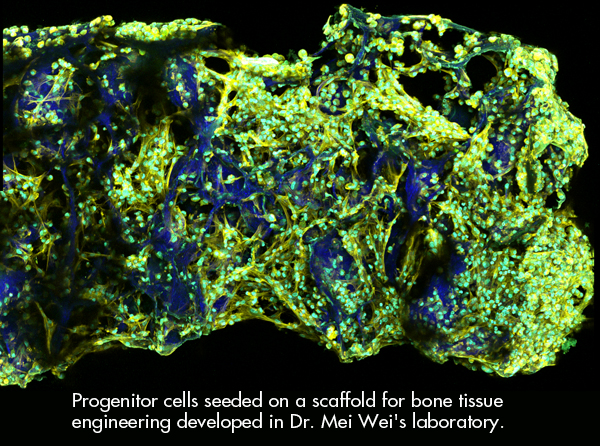 Cell-based bone tissue engineering has the potential to address this shortage of bone graft material, as it employs a biomaterial scaffold carrying progenitor cells that will regenerate new bone at the site of injury. However, numerous obstacles currently prevent the application of cell-based bone tissue engineering. It is not fully understood how the process works, and it is challenging to use this approach to reliably heal large defects. It is also difficult to observe this complex repair process inside a scaffold after it has been implanted.
Cell-based bone tissue engineering has the potential to address this shortage of bone graft material, as it employs a biomaterial scaffold carrying progenitor cells that will regenerate new bone at the site of injury. However, numerous obstacles currently prevent the application of cell-based bone tissue engineering. It is not fully understood how the process works, and it is challenging to use this approach to reliably heal large defects. It is also difficult to observe this complex repair process inside a scaffold after it has been implanted.
In the paper, the authors detail a method for visualizing seeded progenitor and resident host cells as they create new bone inside of a living animal. They use genetically engineered animals that contain bone cells labeled with fluorescent proteins, which allows researchers to track bone cells in the implant. For the group, combining this labeling approach with their model allows the 3D tracking of any protein or cell of interest within a live animal and in a cell-biomaterial construct for tissue engineering. This method is also conducive to tracking the progression of repair in the same animal, allowing researchers to connect the steps of the complex regeneration process and visualize it as it develops over time.
Max Villa (M.S.), lead-author of the paper, is pursuing doctoral research in bone regeneration in the Wei Laboratory. His research focuses on scaffold design and testing as well as two-photon live animal imaging.
He states of the research findings: “Just as the telescope allowed us to see new things for the first time and to ask new questions, this technique allows us to dig deeper into what contributes to successful bone tissue engineering. This methodology should deepen our understanding of this process, ultimately leading to safer and more effective tissue engineering therapies.”
The full article can be accessed at: http://online.liebertpub.com/doi/pdfplus/10.1089/ten.tec.2012.0490.
Published: November 20, 2013
Categories: news, publications, research, undergraduate students
Available Archives